Testicular cancer, although rare in India, with an incidence rate of less than 1 per 100,000, is identified as the most commonly diagnosed tumor among men between the ages of 20-39 years in the world. It is paradoxically the most curable cancer known to medicine with survival rates exceeding 95% and cure rates above 90%. Testicular cancer commonly arises from cells of asexual reproduction and consists of about 95% of testicular neoplasms. It is hence also termed as testicular germ cell tumour (TCGT). TCGT is further categorized into seminoma and nonseminomatous germ cell tumors (NSGCT). The statistics aforementioned are achievable on how early and accurately they are diagnosed.
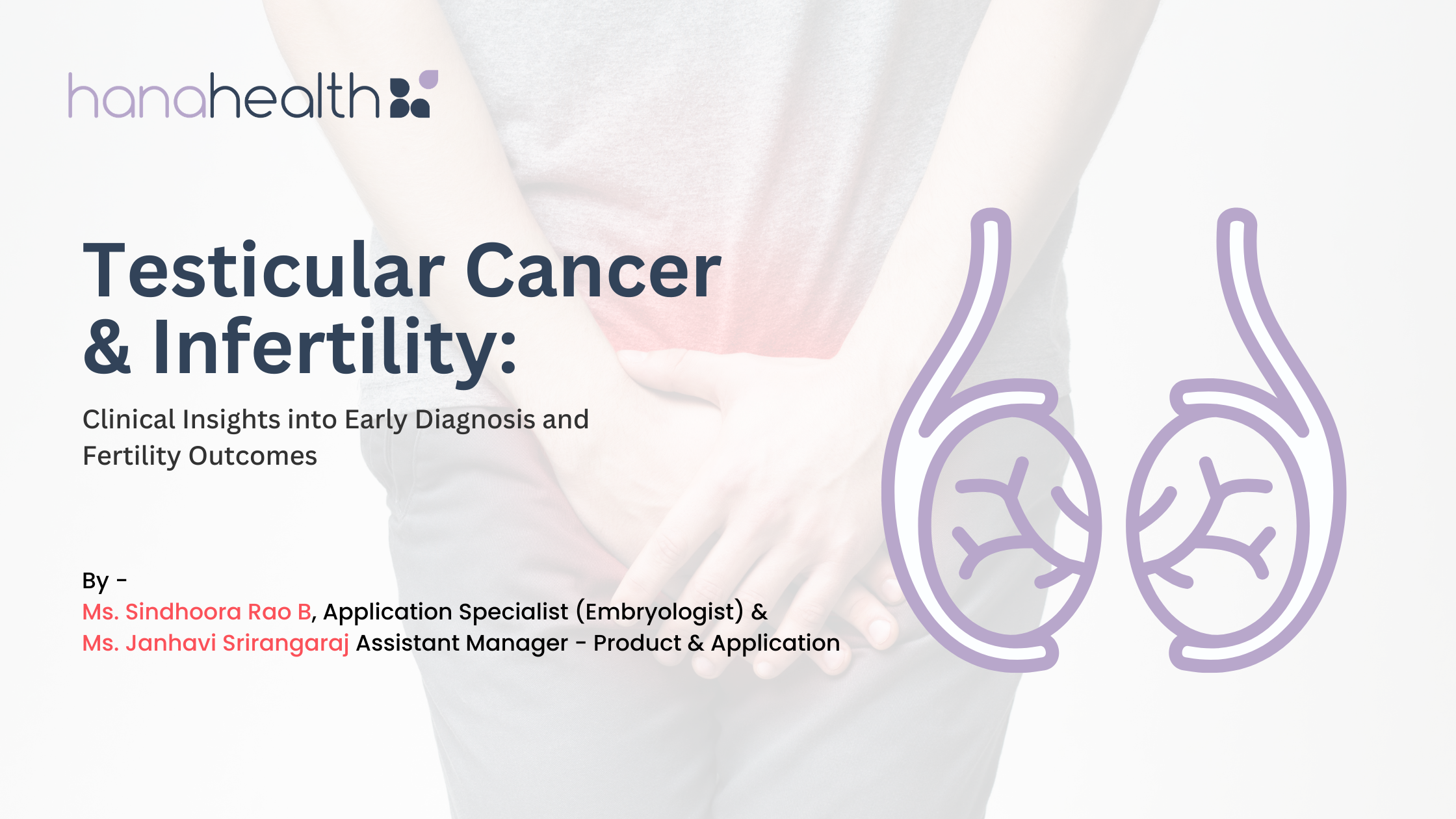
Cross-section diagram of the male reproductive system illustrating the presence of a malignant testicular tumor. Source: https://www.sydneyurologycare.com/testicular-cancer-images
The classical presentation of testicular cancer is a painless, enlarged scrotal mass, occasionally marked by acute pain from rapid intratumoral hemorrhage. Risk factors include cryptorchidism, a personal or family history of testicular cancer, and specific ethnic backgrounds. The most notable problem is delayed diagnosis and more advanced disease at diagnosis, intensified treatment, and greater compromise of the patient’s future fertility. Hence, initial clinical encounters must be treated with absolute diagnostic urgency incorporating physical examination, serum tumor markers, and scrotal ultrasound. At DSS Imagetech, we position ourselves right at this first critical junction. DSS Imagetech supports testicular cancer diagnostics through a comprehensive suite of solutions encompassing next-generation sequencing kits for precise molecular profiling, AI-enhanced digital pathology and laser capture of microdissection platforms for accurate histological evaluation, and advanced imaging and ultrasound technologies for targeted urological assessment.
High-resolution scrotal ultrasound is the first go-to step when evaluating suspected testicular cancer. It is easy to access, completely non-invasive, and cost-effective. When used in the right clinical setting, it has an impressive sensitivity and specificity of over 90%. By using a high-frequency linear transducer, clinicians can easily find out if a problem is inside or outside the testicle. It also helps them see if a lesion is solid or just a fluid-filled cyst, while color Doppler allows them to check the blood flow.
Beyond just looking at the main tumor, ultrasound handles two other crucial jobs. First, it can uncover hidden tumors in patients who already have metastatic disease but seem to have a normal physical exam. These are often referred to as “burned-out” primary tumors. Second, it allows doctors to carefully check the other testicle for additional tumors, even though finding a second one is quite rare.
Sometimes doctors find very small, ambiguous lesions under 10 mm. If the ultrasound is inconclusive and the patient’s blood markers are normal, it is perfectly fine to just keep an eye on it by repeating the ultrasound in six to eight weeks. Newer tools like Contrast-Enhanced Ultrasound (CEUS) and elastography too are stepping up to help. They measure tissue blood flow and stiffness to give doctors a better read on tricky lesions using modern diagnostic machines.
While MRI can be a helpful backup when doctors suspect a mass might actually be benign, it is not always the best tool for the job. It has not proven to be consistently better than a good high-resolution ultrasound, and it relies heavily on having an expert radiologist available to interpret the results correctly.
Testicular cancer is unique because it is one of the few solid organ cancers with reliable serum tumor markers. These markers play a massive role in patient care. Doctors need to measure alpha-fetoprotein (AFP), beta-human chorionic gonadotropin (β-hCG), and lactate dehydrogenase (LDH) before starting any treatment, even before a radical inguinal orchiectomy. Getting these baseline levels is absolutely essential for staging the cancer, assessing risk, tracking how well treatments work, and monitoring for recurrence.
However, clinicians still need to keep the limits of these standard markers in mind. When first diagnosing a patient, the combined sensitivity of these markers is only about 50%. This means a completely normal blood test does not rule out cancer. False positives can also happen. For example, liver disease can raise AFP levels, while hypogonadism or marijuana use can cause spikes in β-hCG. Because of these quirks, the final diagnosis of testicular cancer always comes down to examining the removed tissue under a microscope.
Despite these limitations, blood markers are incredibly valuable for the TNMS staging system. The “S” in TNMS stands for the patient’s serum marker levels after surgery. These levels sort patients into categories from S0 to S3, which directly guides their specific treatment plan. Watching these marker levels drop after treatment is just as important. If the numbers do not fall the way doctors expect, it usually points to leftover cancer or spreading disease.
Some of the most exciting advancements in testicular cancer diagnostics are happening at the molecular level. These breakthroughs are directly changing how clinicians manage patients by offering much greater precision and personalized care.
MicroRNA 371a-3p (miR-371a-3p) is quickly becoming the most promising new biomarker for testicular germ cell tumors. Unlike traditional blood markers, miR-371a-3p shows incredibly high sensitivity for both seminomatous and non-seminomatous types. Most importantly, it avoids the false positives seen with AFP and β-hCG in benign conditions. Several ongoing clinical trials are working to validate its everyday use. For instance, the SWOG S1823 study is investigating whether miR-371a-3p can accurately predict cancer recurrence. Adding this marker to routine surveillance could drastically improve how early doctors catch relapses.
Circulating tumor DNA (ctDNA) represents another major diagnostic frontier. Researchers are actively studying liquid biopsy platforms that detect tumor DNA floating in the bloodstream. These biopsies act as real-time trackers for tumor burden and treatment response. While not yet part of everyday clinical practice for testicular cancer, ctDNA shows incredible potential for monitoring tiny amounts of leftover disease. It could also help identify platinum resistance, which remains a massive clinical hurdle in treating relapsed or stubborn tumors.
We are also gaining a much clearer picture of the genetic architecture behind testicular cancer. Isochromosome 12p, or i(12p), shows up in over 80% of these tumors and acts as a defining genetic hallmark. Clinicians do not rely on FISH testing for i(12p) during standard diagnoses. However, spotting it becomes extremely helpful in ambiguous cases or when tumors appear outside the testicles.
Family history is another vital piece of the puzzle. Men who have a close relative with testicular cancer face a significantly higher risk, and researchers have even linked rare inherited mutations in DNA repair genes like BRCA2 to certain cases. Because of this, genetic counseling is highly recommended for men with a family history, bilateral disease, or very early onset. This counseling is crucial for guiding future surveillance strategies and making informed fertility decisions.
Doctors stage testicular germ cell tumors using the Tumor Node Metastasis Serum marker classification, commonly known as the TNMS system. This framework follows the 8th Edition staging manual published by the American Joint Committee on Cancer. The 8th Edition introduced several important updates that every treating physician needs to know to provide the best care.
First, the manual splits the pathologic T staging for seminomas into pT1a and pT1b categories based on a strict 3 cm size cutoff. This update reflects clinical data showing that larger tumors carry a higher risk of relapse in Stage I seminomas. Interestingly, this specific size rule does not apply to non-seminomatous tumors. Second, the new guidelines expanded the pT2 category. It now includes cases where the tumor invades the hilar soft tissue or epididymis, going well beyond the older criteria of lymphovascular invasion and tunica vaginalis penetration.
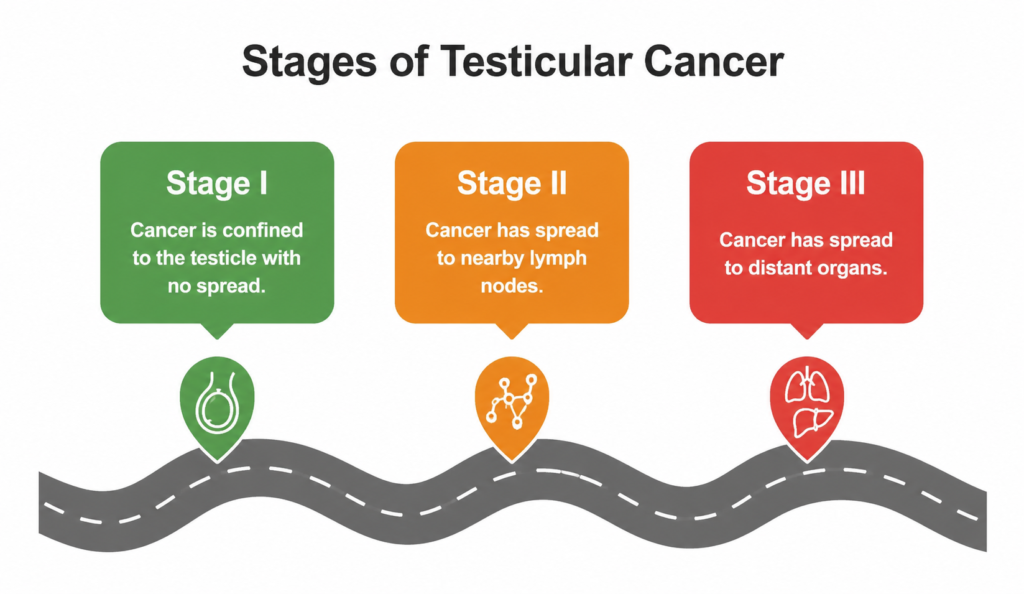
An overview of testicular cancer progression from Stage I to Stage III.
When a patient has Stage I disease, it means the cancer is completely confined to the testicle. Doctors have a few different ways to manage this early stage, such as simply keeping a close watch on it, adding extra chemotherapy, or using radiation. The best approach depends on the specific cell type of the tumor and the patient’s overall risk level.
Stage II disease means the cancer has moved into the lymph nodes in the back of the abdomen. To get a clear picture of what is going on, doctors rely heavily on CT scans of the abdomen and pelvis. If a patient has smaller Stage IIA or IIB seminomas, radiation or chemotherapy usually works well. However, Stage IIC and anything more advanced will require powerful chemotherapy treatments based on cisplatin.
Stage III disease indicates that the cancer has spread to distant parts of the body or that the patient’s blood tumor markers are extremely high. At this point, doctors use comprehensive CT scans of the chest, abdomen, and pelvis. They combine these images with close monitoring of the patient’s blood markers after surgery to figure out exactly how intense the treatment plan needs to be.
Using multiple types of imaging together, like combining a standard ultrasound with CT scans, is the gold standard for both the initial staging and checking how well the cancer responds to treatment
Testicular cancer heavily impacts men in their peak reproductive years, usually between the ages of 20 and 40. Because of this timing, fertility is not just a secondary concern but a core part of the diagnosis. The effects on fertility start much earlier than many doctors realize. In fact, studies reveal that 30 to 50 percent of men with testicular cancer already have poor semen quality at the time of diagnosis before they even begin treatment.
The tumor itself actively disrupts the body’s hormonal balance and increases oxidative stress. It also leads to higher rates of sperm DNA damage and can produce hormones like hCG and AFP that further interfere with sperm production. Hence, the tumor creates a hostile environment for generating sperm. When doctors add treatments like surgery, cisplatin based chemotherapy, radiation, or lymph node removal to an already weakened reproductive system, the combined toll on a patient’s fertility can be massive.
Doctors must discuss and offer sperm cryopreservation at the very time of diagnosis before any therapy starts. Waiting until after treatment is not an option because preservation is a strict pretreatment necessity. For men who have no sperm in their semen at diagnosis, doctors can perform a special extraction procedure during the initial testicle removal surgery. This technique rescues the chance to retrieve sperm before starting therapies that could permanently damage reproductive cells. Recovery of sperm production after treatment is unpredictable. Some men may see improvement within months, while for others it can take several years or may not fully return highlighting the importance of early fertility planning.
In a study by Peluso et al., men with Testicular Cancer made up the largest subgroup among the 254 cancer patients evaluated. Notably, this group showed the poorest semen quality at baseline, including lower sperm concentration, reduced total and progressive motility, decreased viability, and a higher proportion of abnormal sperm forms.
Following semen freezing and thawing, there was a further decline in sperm function. Across the entire study population, recovery rates dropped to about 46.82% for viability and 16.75% for progressive motility. Within the testicular cancer subgroup, these recovery outcomes were even lower, indicating a greater impact on sperm resilience compared to other cancer types.
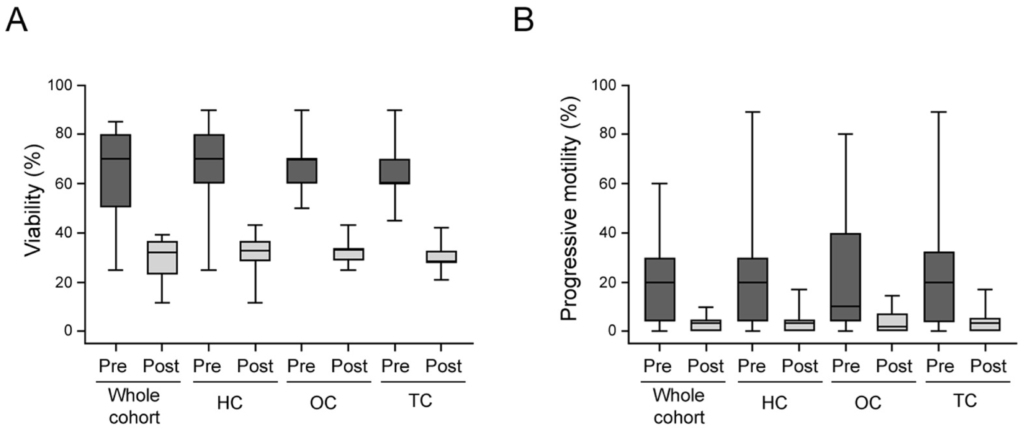
Fig: Box plots of the distribution of pre- (Pre) and post-cryopreservation (Post) for viability (A) and progressive motility (B) stratified by different cancer subgroups. HC, hematological cancers; OC, other cancers; TC, testicular cancer.
In a large Canadian academic fertility centre study (2001 to 2020), a total of 4,521 semen samples were cryopreserved from 2,504 patients. Among these, Testicular Cancer (29.5%) and Lymphoma (26.9%) were the most common diagnoses.
Interestingly, only a small proportion of patients returned to use their stored samples. A total of 81 patients (3.2%) underwent fertility treatment using either Intrauterine Insemination or In Vitro Fertilization, while 62 patients (2.5%) chose to transfer their samples to another clinic.
Looking at outcomes, 67 couples proceeded with treatment, resulting in 66 IVF cycles (104 embryo transfers) and 101 IUI cycles. Among these couples, 53.7% achieved a clinical pregnancy. The success rates differed by method, with about 6.6% per cycle for IUI and 30.8% per embryo transfer for IVF. An interesting finding was that higher sperm concentration or total motile count did not necessarily translate into better pregnancy outcomes. Instead, success was more closely linked to embryo development. Patients who conceived had, on average, nearly two more usable embryos per cycle (1.9 ± 0.8; p = 0.02) compared to those who did not achieve pregnancy.
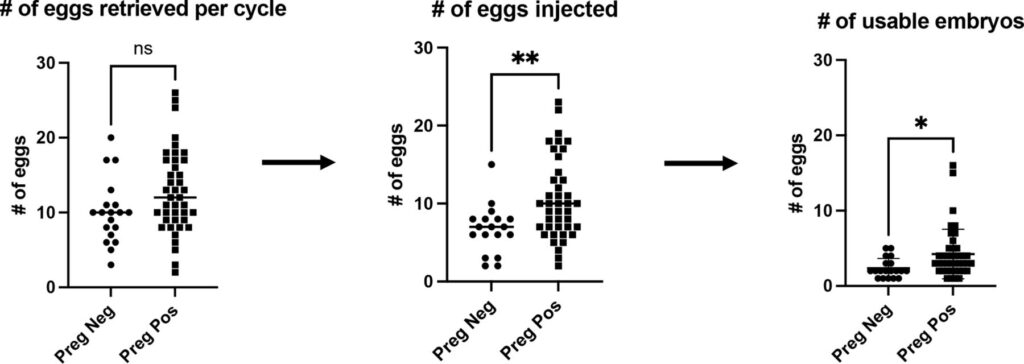
Comparison of the numbers of eggs retrieved for each IVF cycle, the numbers of eggs injected and the number of usable embryos between those who achieved a positive pregnancy (indicated by a positive ß-hCG) and those with a negative pregnancy (negative ß-hCG). IVF, in vitro fertilisation. ** P ≤ 0.01; * P ≤ 0.05
For men diagnosed with Testicular Cancer, parenthood is still very much achievable despite potential impacts on fertility from the disease and its treatment. While sperm production may be reduced or temporarily lost due to surgery, chemotherapy, or radiation, options like sperm cryopreservation before treatment and assisted reproductive techniques such as In Vitro Fertilization and Intracytoplasmic Sperm Injection provide effective pathways to biological fatherhood. Even in cases where fertility is compromised, recovery may occur over time or sperm can be retrieved surgically, and clinical outcomes show meaningful pregnancy success rates among those who pursue treatment. Overall, with timely planning and appropriate medical support, most men with testicular cancer retain a realistic chance of becoming fathers.
Kraft P, Amiri A, Mousa A, Kaushal S, Bacon H, Glicksman RM, Hamilton RJ. Testicular Cancer: Diagnosis, Treatment, and Biomarker Advances. Res Rep Urol. 2026;18:511445 https://doi.org/10.2147/RRU.S511445
Oliveira-Lopes, B., Tavares, N. T., & Lobo, J. (2026). Testicular germ cell tumors and molecular biomarkers. Current opinion in urology, 36(2), 205–211. https://doi.org/10.1097/MOU.0000000000001353
Sykes, J., Kaldany, A., & Jang, T. L. (2024). Current and Evolving Biomarkers in the Diagnosis and Management of Testicular Germ Cell Tumors. Journal of Clinical Medicine, 13(23), 7448. https://doi.org/10.3390/jcm13237448
Valentina Tateo, Zachary J. Thompson, Scott M. Gilbert, Victoria K. Cortessis, Siamak Daneshmand, Timothy A. Masterson, Darren R. Feldman, Phillip M. Pierorazio, Gagan Prakash, Axel Heidenreich, Peter Albers, Andrea Necchi, Philippe E. Spiess, Epidemiology and Risk Factors for Testicular Cancer: A Systematic Review, European Urology, Volume 87, Issue 4, 2025, Pages 427-441,ISSN 0302-2838,
https://doi.org/10.1016/j.eururo.2024.10.023.
https://www.cancer.org/cancer/types/testicular-cancer.html
Parekh R, et al. “Fertility considerations in men with testicular cancer.” Translational Andrology and Urology. 2020.https://doi.org/10.3390/jcm13237448
Peluso, G., Tisato, V., Singh, A. V., Gemmati, D., & Scarpellini, F. (2023). Semen Cryopreservation to Expand Male Fertility in Cancer Patients: Intracase Evaluation of Semen Quality. Journal of personalized medicine, 13(12), 1654. https://doi.org/10.3390/jpm13121654
Chen, T., Hamilton, S., & Liu, K. E. (2024). Twenty-year oncology sperm banking experience at a Canadian academic fertility centre: a retrospective study examining the usage and reproductive outcomes from oncology patients. BMJ open, 14(8), e088112. https://doi.org/10.1136/bmjopen-2024-088112

IVF has helped millions of couples achieve their dream of parenthood, but it’s important to understand that success rates can vary significantly from one case to another. Here are the main factors that influence IVF success.
Read More
The field of In Vitro Fertilization (IVF) has come a long way since the birth of the first IVF baby in 1978. Recent technological .
Read More
When undergoing IVF, many factors can influence your chances of success, and lifestyle choices play a significant role.
Read More